|
|
HOME >>
Pharmaceuticals API List 4 >>
Ceftizoxime Sodium

Ceftizoxime Sodium

CAS Registry Number 68401-82-1
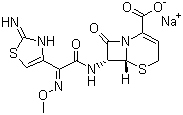
Synonyms
(6R,7R)-7-[[(2Z)-(2-Amino-4-thiazolyl)(methoxyimino)acetyl]amino]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid sodium salt
Molecular Structure Ceftizoxime sodium,
(6R,7R)-7-[[(2Z)-(2-Amino-4-thiazolyl)(methoxyimino)acetyl]amino]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid sodium salt, CAS #: 68401-82-1
Molecular Formula C13H12N5NaO5S2
Molecular Weight 405.38
CAS Registry Number 68401-82-1
ceftizoxime, an antibiotic, to help treat your infection. The drug will
be either injected into a large muscle (such as your buttock or hip) or
added to an intravenous fluid that will drip through a needle or catheter
placed in your vein for 30 minutes, two or three times a day.
Ceftizoxime eliminates bacteria that cause many kinds of infections,
including lung, skin, bone, joint, stomach, blood, and urinary tract
infections. This medication is sometimes prescribed for other uses; ask your
doctor or pharmacist for more information.
a third-generation cephalosporin antibiotic.
indications It is prescribed in the treatment of infections caused by
susceptible bacterial strains (does not include Pseudomonas aeruginosa),
primarily in the respiratory system, genitourinary system, bone, joints, and
skin.
contraindications Known sensitivity to this drug prohibits its use. It is
used with caution in patients who are allergic to penicillin.
Side effects Return to top
Ceftizoxime may cause side effects. If you are administering ceftizoxime
into a muscle, it may be mixed with lidocaine (Xylocaine) to reduce pain at
the injection site. Tell your health care provider if any of these symptoms
are severe or do not go away:
* diarrhea
* stomach pain
* upset stomach
* vomiting
If you experience any of the following symptoms, call your health care
provider immediately:
* unusual bleeding or bruising
* difficulty breathing
* itching
* rash
* hives
* sore mouth or throat
Dosage and Administration
Adults
IV/IM 1 to 2 g every 8 to 12 h (life-threatening infections: IV up to 2 g
every 4 h or 3 to 4 g every 8 h).
Children older than 6 mo of age
IV/IM 50 mg/kg every 6 to 8 h up to 200 mg/kg/day (max, 12 g/day).

|


