|
|
HOME >>
API >>
Nitroglycerin
Nitroglycerin [CAS 55-63-0]

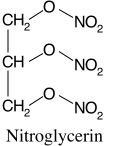
IUPAC Name:
1,3-dinitrooxypropan-2-yl nitrate | CAS Registry Number: 55-63-0
Synonyms: nitroglycerin, Nitrostat,
Trinitroglycerin, Glyceryl trinitrate, Nitroglycerine, Nitroglycerol,
Nitroderm, Nitroglyn, Nitrospan, Minitran, Nitro-dur, Nitrol,
Trinitroglycerol, Perlinganit, Epinitril, Gilustenon, Millisrol, Nitrocine,
Nitromint, Nitroplast
|
Molecular Formula:
|
C3H5N3O9 |
Molecular Weight:
|
227.086500 [g/mol] |
|
H-Bond Donor:
|
0 |
H-Bond Acceptor:
|
9 |
Exposure Limits
NIOSH REL: ST 0.1 mg/m3 [skin]
OSHA PEL†: C 0.2 ppm (2 mg/m3) [skin]
InChIKey:
SNIOPGDIGTZGOP-UHFFFAOYSA-N
Nitroglycerin Property
Fp : 12°C
CAS Data Base Reference: 55-63-0(CAS DataBase Reference)
NIST Chemistry Reference: 1,2,3-Propanetriol, trinitrate(55-63-0)
EPA Substance Registry System: 1,2,3-Propanetriol,
trinitrate(55-63-0)
Safety
Hazard Codes : E,T+,N
Risk Statements : 11-51/53-33-26/27/28-3
Safety Statements : 7-16-61-45-36/37-35-33
RIDADR : 1993
Hazard
Class : 1.1A
Packing Group : I
Hazardous Substances Data: 55-63-0(Hazardous Substances Data)
Nitroglycerin Chemical Properties, Usage, Production
General Description:
Colorless to pale-yellow, viscous liquid or solid (below 56°F). (Note: An
explosive ingredient in dynamite (20-40%) with ethylene glycol dinitrate
(80-60%).).
Air & Water Reactions
Highly flammable.
Reactivity Profile
Nitroalkanes,
Packing: N/A
Explosive Liquid
Incompatibilities & Reactivities ::
Heat, ozone, shock, acids [Note: An OSHA Class A Explosive (1910.109).]
Reactivity Profile:
Nitroalkanes, such as NITROGLYCERIN, range from slight to strong oxidizing
agents. If mixed with reducing agents, including hydrides, sulfides and
nitrides, they may begin a vigorous reaction that culminates in a
detonation. Nitroalkanes are milder oxidizing agents, but still react
violently with reducing agents at higher temperature and pressures.
Nitroalkanes react with inorganic bases to form explosive salts. The
presence of metal oxides increases the thermal sensitivity of nitroalkanes.
Nitroalkanes with more than one nitro group are generally explosive.
Nitroalkanes are insoluble in water. Flammable/combustible material. May be
ignited by heat, sparks or flames. Nitroglycerin is incompatible with the
following: Heat, ozone, shock, acids. Note: An OSHA Class A Explosive
(1910.109). .
Health Hazard:
Inhalation or contact with material may irritate or burn skin and eyes. Fire
may produce irritating, corrosive and/or toxic gases. Vapors may cause
dizziness or suffocation. Runoff from fire control may cause pollution.
Fire Hazard
HIGHLY
FLAMMABLE: Will be
easily ignited by heat, sparks or flames. Vapors may form explosive mixtures
with air. Vapors may travel to source of ignition and flash back. Most
vapors are heavier than air. They will spread along ground and collect in
low or confined areas (sewers, basements, tanks). Vapor explosion hazard
indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion
hazard. Containers may explode when heated. Many liquids are lighter than
water.
**** [Note: An explosive ingredient in dynamite (20-40%) with ethylene
glycol dinitrate (80-60%).]

|
 |
 |
|
|
PDF DOWNLOAD |
WORD DOCUMENT |
|
|
|
|
|
|
|
|
Product Enquiry |
Product Quotation |
Sample Request |
Place Your Order |
|
|
|
|
|
|
|
|
Note: **** These chemicals are designated as
those that are used in the manufacture of the controlled substances
and are important to the manufacture of the substances. For any
(Control Substance) products Import and Export *** subjected to your
country government laws /control substance ACT.
Note /Government Notification: These chemicals are designated as
those that are used in the manufacture of the controlled substances
and are important to the manufacture of the substances. For any
(Control Substance) products Import and Export *** subjected to your
country government laws /control substance ACT.
Information: The information on this web page is provided to help
you to work safely, but it is intended to be an overview of hazards,
not a replacement for a full Material Safety Data Sheet (MSDS). MSDS
forms can be downloaded from the web sites of many chemical
suppliers. ,also that the information on the PTCL Safety web site,
where this page was hosted, has been copied onto many other sites,
often without permission. If you have any doubts about the veracity
of the information that you are viewing, or have any queries, please
check the URL that your web browser displays for this page. If the
URL begins "www.tajapi.com/www/Denatonium Benzoate.htm/" the page is
maintained by the Safety Officer in Physical Chemistry at Oxford
University. If not, this page is a copy made by some other person
and we have no responsibility for it.
The Controlled Substances Act (CSA) was enacted into law by the
Congress of the United States as Title II of the Comprehensive Drug
Abuse Prevention and Control Act of 1970.[1] The CSA is the federal
U.S. drug policy under which the manufacture, importation,
possession, use and distribution of certain substances is regulated.
The Act also served as the national implementing legislation for the
Single Convention on Narcotic Drugs |
|

|






