|
HOME >>
Chemicals
>>
Chemicals List 2 >> Diethyl
ether
Diethyl ether
CAS number 60-29-7
Identification
Synonyms: Ether; ether,anhydrous; Diethyl ether;
1,1'Oxybisethane;ethyl oxide; diethyl oxide; Ethyl ether anhydrous;
CAS No.: 60-29-7
Molecular Weight: 74.12
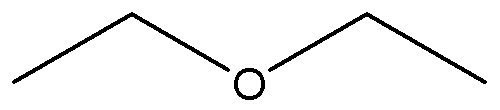
Chemical Formula: C2H5OC2H5
EC No: 200-467-2
Annex I Index No: 603-022-00-4
Physical data
Appearance: colourless liquid
Melting point: -116 C
Boiling point: 34.6 C
Specific gravity: 0.71
Vapour pressure: 400 mm Hg at 18 C
Flash point: -40 C
Explosion limits: 1.7% - 48%
Autoignition temperature: 170 C
Water solubility: 6.9% (20 C)
Stability
Stable, but light-sensitive, sensitive to air. May contain BHT
(2,6-di-tert-butyl-4-methylphenol) as a stabilizer. Substances to be
avoided include zinc, halogens, halogen-halogen compounds,
nonmetals, nonmetallic oxyhalides, strong oxidizing agents, chromyl
chloride, turpentine oils, turps substitutes, nitrates, metallic
chlorides.
Extremely flammable. This material is a serious fire and explosion
risk. Vapour may travel considerable distances to ignition sources,
which need not be an open flame, but may be a hot plate, steam pipe,
etc. Vapour may be ignited by the static electricty which can build
up when ether is being poured from one vessel into another. If large
quantities of material are being poured, suitable precautions must
be taken.
May form explosive peroxides on storage. Peroxides are generally
higher-boiling than the materials from which they form.
Consequently, if a peroxide-containing solution is heated, the
residue becomes progressively more concentrated in peroxide and the
risk of explosion increases rapidly. Vapour-air mixture explosive
(note wide explosion limits). As at 6.11.97 regulated in the UK
under the Highly Flammable Liquids and Liquefied Petroleum Gases
Regulations 1972.
Toxicology
Harmful by ingestion, inhalation or through skin contact. May cause
inebriation or coma. May cause allergic reaction. Skin, eye and
respiratory system irritant. Typical 8h TWA 400 ppm.
Toxicity data
ORL-MAN LDLO 260 mg kg-1
ORL-RAT LD50 1215 mg kg-1
IHL-MUS LC50 31000 ppm/30m Irritation data EYE-HMN 100 ppm
SKN-RBT 360 mg open mld
SKN-GPG 50 mg/24h sev Risk phrases R12 R19 R22 R66 R67.
Personal protection
Remove any source of ignition from the working area, including
hot plates, bunsen burners, hot air guns and electrical equipment.
Only work in very well ventilated areas.
Wear safety glasses.
If using ether from a container that has been open for some time,
test the liquid for the presence of peroxides before use.
Store under an inert atmosphere.
Disposal
Do not attempt to flush this material down a sink. Dangerous levels
of vapour can build up in the sink or within sewers. Store in a
waste solvent container for disposal.
Protective equipment
Safety glasses. Diethyl ether may have a defatting effect on the
skin. If gloves are required, polyvinyl alcohol (PVA) is suggested.
Diethyl ether, also known as ether and ethoxyethane, is a clear,
colorless, and highly flammable liquid with a low boiling point and
a characteristic odor. It is the most common member of a class of
chemical compounds known generically as ethers.
It is an isomer of butanol. Diethyl ether has the formula
CH3-CH2-O-CH2-CH3.
It is used as a common solvent and has been used as a general
anesthetic. Ether is sparingly soluble in water (6.9 g/100 mL). The
diffusion of diethyl ether in air is 0.918*10^-5 m2/s (298K 101.325
kPa).
Diethyl ether is a great solvent for many things, but is extremely
flammable. Professional chemists will be well appraised of the
hazards presented in using ether, but the layperson is less likely
to be aware of these dangers. Diethyl Ether vapors 'hug' the ground,
and in dry air explosive peroxides can form. In other words, even in
a spark/flame free environment, explosions can still happen when
ether vapour is encountered.
For this reason its probably a good idea to have some way of
removing vapours from the vicinity (a fume hood would be a fine
example) and (Zaphraud suggests) one should not use ether on days
with extremely low humidity. Because diethyl ether is so flammable,
and prone to ingition, this procedure should be carried out using a
hotplater/stirrer designed for use in flammable environments. Such a
heater/stirrer does not produce a contact spark when the hotplate is
turned on, and generally employs a brushless AC motor for the
stirrer, because DC motors with brushes generally produce small
sparks which could ignite any stray vapours.
Diethyl ether is prepared from ethanol (a.k.a grain alcohol, ethyl
alcohol, drinking alcohol) by heating it with concentrated. The
reaction proceeds thru an intermediary, "Ethyl sulfuric acid", as do
most reactions of this type.
| |
|
Note /Government
Notification: These chemicals are designated as those that are
used in the manufacture of the controlled substances and are
important to the manufacture of the substances. For any (Control
Substance) products Import and Export *** subjected to your
country government laws /control substance ACT.
Information: The information on this web page is provided to
help you to work safely, but it is intended to be an overview of
hazards, not a replacement for a full Material Safety Data Sheet
(MSDS). MSDS forms can be downloaded from the web sites of many
chemical suppliers. ,also that the information on the PTCL
Safety web site, where this page was hosted, has been copied
onto many other sites, often without permission. If you have any
doubts about the veracity of the information that you are
viewing, or have any queries, please check the URL that your web
browser displays for this page. If the URL begins "www.tajapi.com/www/Denatonium
Benzoate.htm/" the page is maintained by the Safety Officer in
Physical Chemistry at Oxford University. If not, this page is a
copy made by some other person and we have no responsibility for
it.
The Controlled Substances Act (CSA) was enacted into law by the
Congress of the United States as Title II of the Comprehensive
Drug Abuse Prevention and Control Act of 1970.[1] The CSA is the
federal U.S. drug policy under which the manufacture,
importation, possession, use and distribution of certain
substances is regulated. The Act also served as the national
implementing legislation for the Single Convention on Narcotic
Drugs |
|
|
New Chemicals

Diethyl ether,
Potassium permanganate,
Toluene,
Diethyl ether,
Potassium permanganate,
Methyl isobutyl ketone,
Para Anisyl Choride,
Methyl Tyramine,
Para
Cresyl Acetate,
Anisyl Formate,
Ortho Anisic Aldehyde,
3-Benzyloxy Propionitrile,
Veratrol,
Manganese Sulphate Monohydrate
|



