|
|
HOME >>
Pharmaceuticals API List 4 >>
Cefotaxime Sodium

Cefotaxime Sodium

CAS Registry Number is 64485-93-4.
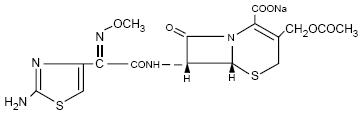
cefotaxime sodiumis a semisynthetic, broad spectrum cephalosporin
antibiotic for parenteral administration. It is the sodium salt of
7-[2-(2-amino-4-thiazolyl) glyoxylamido]-3-(hydroxymethyl)-8-oxo-5-thia-1-azabicyclo
[4.2.0] oct-2-ene-2-carboxylate 72 (Z)-(o-methyloxime), acetate (ester).
CLAFORAN contains approximately 50.5 mg (2.2 mEq) of sodium per gram of
cefotaxime activity. Solutions of CLAFORAN range from very pale yellow to
light amber depending on the concentration and the diluent used. The pH of
the injectable solutions usually ranges from 5.0 to 7.5.
The CAS Registry Number is 64485-93-4.
Indications and Usage
Treatment of infections of lower respiratory tract including pneumonia,
urinary tract, skin and skin structures, bone and joints; treatment of
bacteremia/septicemia, CNS infections, intra-abdominal infections including
peritonitis, gynecological infections including pelvic inflammatory disease,
endometritis and pelvic cellulitis caused by susceptible strains of specific
microorganisms; perioperative prophylaxis.
Side effects
Cefotaxime may cause side effects. If you are administering cefotaxime into
a muscle, it may be mixed with lidocaine (Xylocaine) to reduce pain at the
injection site. Tell your health care provider if any of these symptoms are
severe or do not go away:
* diarrhea
* stomach pain
* upset stomach
* vomiting
If you experience any of the following symptoms, call your health care
provider immediately:
* unusual bleeding or bruising
* difficulty breathing
* skin rash
* itching
* hives
* sore mouth or throat
Storage/Stability/Compatibility -
Cefotaxime sodium sterile powder for injection should be stored at
temperatures of less than 30°C; protect from light. The commercially
available frozen injection should be stored at temperatures no greater than
-20°C. Depending on storage conditions, the powder or solutions may darken
which may indicate a loss in potency.
Doses -
Horses:
For susceptible infections:
a) Foals: 20 - 30 mg/kg IV q6h (Caprile and Short 1987)
Dosage Forms/Preparations/FDA Approval Status -
Veterinary-Approved Products: None
Human-Approved Products:
Cefotaxime Sodium Powder for Injection; 500 mg, 1 g (as cefotaxime), 2 g, 10
g;
Cefotaxime Sodium for Injection in 5% dextrose bags (50 ml)—frozen; 1 g, 2
g;
|


