|
HOME >>
API >>
Ephedrine Hydrochloride USP


|
Ephedrine Hydrochloride USP

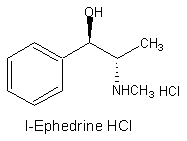 |
Molecular Formula :
C10H15NO. HCl
Molecular Weight :
201.69
CAS No. :
[50-98-6]
Chemical Name :
1. Benzenemethanol,
œ-[1-(methylamino)ethyl]-, hydrochloride [R-(R*,S* )], salt
2.
(1R,2S)-2-Methylamino-1-phenyl propan-1-ol Hydrochloride
Specifications :
| 1. |
Description : |
Colourless
crystals or white crystalline powder |
| 2. |
Identification : |
a) Infrared
spectrum of sample is concordant with the reference
spectrum.
b) A solution of sample respond to the test for chloride. |
| 3. |
Solubility : |
Freely
soluble in water and soluble in alcohol. |
| 4. |
Melting range : |
Between 217°
and 220°C; the range between beginning and end of melting
does not exceed 2°C |
| 5. |
Specific rotation : |
Between -
33.0° and - 35.5° ( 5 % solution in water ) |
| 6. |
Acidity & alkalinity : |
NMT 0.10 ml
of 0.02 N H2SO4 or NMT 0.20 ml of 0.02 N
NaOH solution |
| 7. |
Sulfate : |
Must pass the
test. No turbidity develops within 10 min. |
| 8. |
Ordinary impurities : |
NMT 2.0 % |
| 9. |
Residue on ignition : |
NMT 0.1 % |
| 10. |
Loss
on drying : |
NMT 0.5 % |
| 11. |
Organic volatile impurities : |
Must pass the test
|
| 12. |
Assay
: |
Between 98.0 % and 100.5 % (
dried basis ) |
Therapeutic indications :
Bronchodilator
Packing :
Packed in double lined polyethylene bags in 25 kg
fiber drums.
Storage :
Store below 35°C. |
 Ephedrine
(EPH) is a sympathomimetic amine commonly used as a stimulant,
appetite suppressant, concentration aid, decongestant, and to treat
hypotension associated with anaesthesia. Ephedrine is similar in
structure to the synthetic derivatives amphetamine and
methamphetamine. Chemically, it is an alkaloid derived from various
plants in the genus Ephedra (family Ephedraceae). It is most usually
marketed in the hydrochloride and sulfate forms. Ephedrine
(EPH) is a sympathomimetic amine commonly used as a stimulant,
appetite suppressant, concentration aid, decongestant, and to treat
hypotension associated with anaesthesia. Ephedrine is similar in
structure to the synthetic derivatives amphetamine and
methamphetamine. Chemically, it is an alkaloid derived from various
plants in the genus Ephedra (family Ephedraceae). It is most usually
marketed in the hydrochloride and sulfate forms.
Chemistry
Ephedrine exhibits optical isomerism and has two chiral centres. By
convention the enantiomers with opposite stereochemistry around the
chiral centres are designated ephedrine, while pseudoephedrine has
same stereochemistry around the chiral carbons. That is, (1R,2R)-
and (1S,2S)-enantiomers are designated pseudoephedrine; while
(1R,2S)- and (1S,2R)-enantiomers are designated ephedrine.
The isomer which is marketed is (-)-(1R,2S)-ephedrine.
As with other phenylethylamines, it is also somewhat chemically
similar to methamphetamine, although the amphetamines are more
potent and have additional biological effects.
These terms are used to refer to the same substance derived from the
plant Ephedra. (There are many common names for these evergreen
plants, including squaw tea and Mormon tea.) Ephedra is a shrub-like
plant that is found in desert regions in central Asia and other
parts of the world. The dried greens of the plant are used
medicinally. Ephedra is a stimulant containing the herbal form of
ephedrine, an FDA-regulated drug found in over-the-counter asthma
medications.
In the United States, ephedra and ephedrine are sold in health food
stores under a variety of brand names. Ephedrine is widely used for
weight loss, as an energy booster, and to enhance athletic
performance. These products often contain other stimulants, such as
caffeine, which may have synergistic effects and increase the
potential for adverse effects. Ephedra is often touted as the
"herbal fen-phen."
Ephedra's main active medical ingredients are the alkaloids
ephedrine and pseudoephedrine. The ephedras also contain various
tannins and related chemicals.The stem contains 1-3% total
alkaloids, with ephedrine accounting for 30-90% of this total. The
concentrations of these alkaloids depends upon the particular
species of ephedra used.
Clinical use
Ephedrine Sulphate (1932) Ephedrine Compound (1932) and Swan-Myers
Ephedrine Inhalant No. 66 (ca. 1940)
Indications
In traditional Chinese medicine, ephedrine has been used in the
treatment of asthma and bronchitis for centuries.
An ECA stack is a component found in thermogenic weight loss pills,
composed of ephedrine, caffeine and aspirin (many supplement
manufacturers include salicin instead of aspirin) working to speed
up the metabolism and thus cause food energy to burn faster. The ECA
stack is a popular supplement taken by body builders before workouts
due to the increased amount of energy and alertness.
Recreational and illicit use
Anecdotal reports have suggested that ephedrine helps studying,
thinking, or concentrating to a greater extent than caffeine. Some
students and some white-collar workers have used ephedrine (or
Ephedra-containing herbal supplements) for this purpose, as well as
some professional athletes and weightlifters. It is common for many
athletes to use stimulants while exercising. Such use of ephedrine
has been associated with stimulant dependence, as well as deaths
from heatstroke in athletes and circulatory problems such as aortic
aneurysm in weightlifters, though these side effects are rare.As a
phenylethylamine, ephedrine has a similar chemical structure to
amphetamines. Ephedrine can be used in the synthesis of
methamphetamine by chemical reduction; this has made ephedrine a
highly sought-after chemical precursor in the illicit manufacture of
methamphetamine. The most popular method for reducing ephedrine to
methamphetamine is similar to the Birch reduction, in that it uses
anhydrous ammonia and lithium metal in the reaction. The second most
popular method uses red phosphorus, iodine, and ephedrine in the
reaction.
 Note:
These API/ chemicals are designated as those that are used in
the manufacture of the controlled substances and are important to
the manufacture of the substances. For any (Control Substance)
products Import and Export *** subjected to your country government
laws /control substance ACT. Note:
These API/ chemicals are designated as those that are used in
the manufacture of the controlled substances and are important to
the manufacture of the substances. For any (Control Substance)
products Import and Export *** subjected to your country government
laws /control substance ACT.
Note /Government Notification:
These chemicals are designated as those that are used in the
manufacture of the controlled substances and are important to the
manufacture of the substances. For any (Control Substance) products
Import and Export *** subjected to your country government laws
/control substance ACT.
Information: The information on this web page is provided to
help you to work safely, but it is intended to be an overview of
hazards, not a replacement for a full Material Safety Data Sheet (MSDS).
MSDS forms can be downloaded from the web sites of many chemical
suppliers. ,also that the information on the PTCL Safety web site,
where this page was hosted, has been copied onto many other sites,
often without permission. If you have any doubts about the veracity
of the information that you are viewing, or have any queries, please
check the URL that your web browser displays for this page. If the
URL begins "www.tajapi.com/www/Denatonium Benzoate.htm/" the page is
maintained by the Safety Officer in Physical Chemistry at Oxford
University. If not, this page is a copy made by some other person
and we have no responsibility for it.
The Controlled Substances Act (CSA) was enacted into law by the
Congress of the United States as Title II of the Comprehensive Drug
Abuse Prevention and Control Act of 1970.[1] The CSA is the federal
U.S. drug policy under which the manufacture, importation,
possession, use and distribution of certain substances is regulated.
The Act also served as the national implementing legislation for the
Single Convention on Narcotic Drugs.
|
![Ephedrine CAS No. : [50-98-6]](new%20update1/ephedrak.jpg)
Also Available:
| (l) - Ephedrine Sulphate |
[ 134 - 72 - 5 ] |
| (l) - Ephedrine Base Anhydrous |
[ 299 - 42 - 3 ] |
| (l) - Ephedrine Base
Hemihydrate |
[ 50906 - 05 - 3 ] |
| (d) - Ephedrine Hydrochloride |
[ 24221 - 86 - 1 ] |
| (d) - Ephedrine Base
Hemihydrate |
[ 144429 - 10 - 7 ]
|
|






 Ephedrine
(EPH) is a sympathomimetic amine commonly used as a stimulant,
appetite suppressant, concentration aid, decongestant, and to treat
hypotension associated with anaesthesia. Ephedrine is similar in
structure to the synthetic derivatives amphetamine and
methamphetamine. Chemically, it is an alkaloid derived from various
plants in the genus Ephedra (family Ephedraceae). It is most usually
marketed in the hydrochloride and sulfate forms.
Ephedrine
(EPH) is a sympathomimetic amine commonly used as a stimulant,
appetite suppressant, concentration aid, decongestant, and to treat
hypotension associated with anaesthesia. Ephedrine is similar in
structure to the synthetic derivatives amphetamine and
methamphetamine. Chemically, it is an alkaloid derived from various
plants in the genus Ephedra (family Ephedraceae). It is most usually
marketed in the hydrochloride and sulfate forms.





![Ephedrine CAS No. : [50-98-6]](new%20update1/ephedrak.jpg)