|
HOME >>
API >>
Phenylephrine

Systematic (IUPAC) name

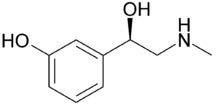
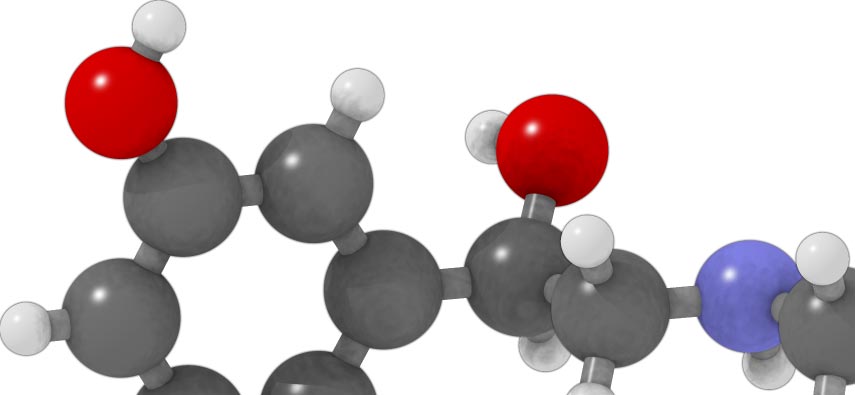
(R)-3-[-1-hydroxy-2-(methylamino)ethyl]phenol
 Phenylephrine
or Neo-Synephrine is an α1-adrenergic receptor agonist used
primarily as a decongestant, as an agent to dilate the pupil,
and to increase blood pressure. Phenylephrine has recently been
marketed as a substitute for pseudoephedrine but there are recent
claims that oral phenylephrine may be no more effective as a
decongestant than a placebo. Phenylephrine
or Neo-Synephrine is an α1-adrenergic receptor agonist used
primarily as a decongestant, as an agent to dilate the pupil,
and to increase blood pressure. Phenylephrine has recently been
marketed as a substitute for pseudoephedrine but there are recent
claims that oral phenylephrine may be no more effective as a
decongestant than a placebo.
Chemical data
Formula : C9H13NO2
Mol. mass : 167.205 g/mol
SMILES : eMolecules
 Pharmacokinetic
data Pharmacokinetic
data
Bioavailability 38% through GI tract
Protein binding 95%
Metabolism Hepatic (monoamine oxidase)
Half life 2.1 to 3.4 hours
Routes Oral, intranasal, ophthalmic, intravenous, intramuscular
Phenylephrine is used to relieve nasal
discomfort caused by colds, allergies, and hay fever. It is also
used to relieve sinus congestion and pressure. Phenylephrine will
relieve symptoms but will not treat the cause of the symptoms or
speed recovery. Phenylephrine is in a class of medications called
nasal decongestants. It works by reducing swelling of the blood
vessels in the nasal passages.
Uses
Phenylephrine comes as a tablet, a liquid, or a dissolving strip to
take by mouth. It is usually taken every 4 hours as needed.
Follow the directions on your prescription label or the package
label carefully, and ask your doctor to explain any part you do not
understand. Take phenylephrine exactly as directed.
Phenylephrine comes alone and in combination with other medications.
Check nonprescription cough and cold product labels carefully before
using two or more products at the same time. These products may
contain the same active ingredient(s) and taking them together could
cause you to receive an overdose. This is especially important if
you will be giving cough and cold medications to a child.
If you are giving phenylephrine or a combination product that
contains phenylephrine to a child, read the package label carefully
to be sure that it is the right product for a child of that age. Do
not give phenylephrine products that are made for adults to
children.
Before you give a phenylephrine product to a child, check the
package label to find out how much medication the child should
receive. Give the dose that matches the child's age on the chart.
Ask the child's doctor if you don't know how much medication to give
the child.
Phenylephrine may cause side effects.
* nervousness
* dizziness
* sleeplessness
Phenylephrine is sometimes used as a vasopressor to increase the
blood pressure in unstable patients with hypotension. Such use
is more common in anesthesia or critical-care practices;
phenylephrine is especially useful in counteracting the hypotensive
effect of epidural and subarachnoid anesthetics. It also has the
advantage of not being inotropic or chronotropic, and so it strictly
elevates the blood pressure without increasing the heart rate or
contractility (reflex bradycardia may result from the blood pressure
increase, however). This is especially useful if the heart is
already tachycardic and/or has a cardiomyopathy. The elimination
half life of phenylephrine is about 2.5 to 3 hours.
Substitute for pseudoephedrine
Pseudoephedrine and phenylephrine are both used as decongestants;
and, until recently, pseudoephedrine was much more commonly
available in the United States. This has changed because provisions
of the Combat Methamphetamine Epidemic Act of 2005 placed
restrictions on the sale of pseudoephedrine products in order to
prevent the clandestine manufacture of methamphetamine. Since 2004,
phenylephrine has been increasingly marketed as a substitute for
pseudoephedrine; some manufacturers have changed the active
ingredients of products to avoid the restrictions on sales.
Phenylephrine has been off patent for some time, and there are many
generic brands available.
Clinical Pharmacology
Phenylephrine is sympathomimetic vasoconstrictor that has been used
as a nasal decongestant for many years . Phenylephrine has one
chiral centre and can exist as either the S(+) or R(-) enantiomer.
The R(-) enantiomer is the one used in products containing
phenylephrine. It is a relatively selective alpha-adrenoceptor
agonist. The majority of the sympathomimetic action is due to direct
stimulation of the adrenoceptors and relatively little is due to an
indirect effect via release of noradrenaline [1]. Its pressor action
is weaker than that of noradrenaline but of longer
duration [4]. At therapeutic doses, it does not cause significant
stimulation of the central nervous system.
Sympathomimetic decongestants reduce the nasal congestion due to
increased nasal blood flow associated with colds and influenza. This
effect forms the therapeutic basis for their use in these
conditions.
Hypertensive patients should be aware of the possible side effects
of nonprescription medications on blood pressure control. For
absolute safety no adrenergic agents should be used. However, when
required, phenylephrine is the safest of these agents. Studies
assessing the hypertensive effect of oral phenylephrine in
normotensive volunteers have demonstrated that the minimal dose
required to elicit an increase in blood pressure is approximately 50
mg that is five times the therapeutic dose.
Doses in excess of 120 mg are required to elicit a significant
effect on blood pressure. A recent study in normotensive volunteers
demonstrated that following administration of a cold relief product
containing phenylephrine 10 mg and caffeine 60 mg, there was a small
but statistically significant increase in total peripheral
resistance but no consistent effect on other cardiovascular
parameters including heart rate and blood
pressure.
Interactions
The coadministration of Monoamine Oxidase Inhibitors (MAOIs) or
tricyclic antidepressants and an indirect or mixed-acting
sympathomimetic may result in a hypertensive crisis. Direct-acting
sympathomimetics appear to interact minimally, if at all [16 - Drug
Interaction Facts. 4th edition]. Such concomitant use is clearly
identified as a contra-indication on the labelling of all
phenylephrine-containing products and the appropriate warnings are
provided. Additionally sympathomimetics may reduce the efficacy of
beta-blocking and anti-hypertensive drugs. Conditions where these
drugs are used are contra-indicated for the product.
Storage/Stability/Compatibility
The injectable product should be stored protected from light. Do not
use solutions if they are brown or contain a precipitate. Oxidation
of the drug can occur without a color change. To protect against
oxidation, the air in commercially available ampules for injection
is replaced with nitrogen and a sulfite added.
Phenylephrine is reported to be compatible with all commonly used
IV solutions and the following drugs: chloramphenicol sodium
succinate, dobutamine HCl, lidocaine HCl, potassium chloride, and
sodium bicarbonate. While stated to be incompatible with alkalies,
it is stable with sodium bicarbonate solutions. Phenylephrine is
reported to be incompatible with ferric salts, oxidizing agents,
and metals.
 Note:
These API/ chemicals are designated as those that are used in
the manufacture of the controlled substances and are important to
the manufacture of the substances. For any (Control Substance)
products Import and Export *** subjected to your country government
laws /control substance ACT. Note:
These API/ chemicals are designated as those that are used in
the manufacture of the controlled substances and are important to
the manufacture of the substances. For any (Control Substance)
products Import and Export *** subjected to your country government
laws /control substance ACT.
Note /Government Notification:
These chemicals are designated as those that are used in the
manufacture of the controlled substances and are important to the
manufacture of the substances. For any (Control Substance) products
Import and Export *** subjected to your country government laws
/control substance ACT.
Information: The information on this web page is provided to
help you to work safely, but it is intended to be an overview of
hazards, not a replacement for a full Material Safety Data Sheet (MSDS).
MSDS forms can be downloaded from the web sites of many chemical
suppliers. ,also that the information on the PTCL Safety web site,
where this page was hosted, has been copied onto many other sites,
often without permission. If you have any doubts about the veracity
of the information that you are viewing, or have any queries, please
check the URL that your web browser displays for this page. If the
URL begins "www.tajapi.com/www/Denatonium Benzoate.htm/" the page is
maintained by the Safety Officer in Physical Chemistry at Oxford
University. If not, this page is a copy made by some other person
and we have no responsibility for it.
The Controlled Substances Act (CSA) was enacted into law by the
Congress of the United States as Title II of the Comprehensive Drug
Abuse Prevention and Control Act of 1970.[1] The CSA is the federal
U.S. drug policy under which the manufacture, importation,
possession, use and distribution of certain substances is regulated.
The Act also served as the national implementing legislation for the
Single Convention on Narcotic Drugs.

>>
New Product
Introduced :
Oseltamivir
Phosphate,
Phenyl Propanolamine,
Phenylephrine,
Etafedrine
|

Phenylephrine Meta-analysis
suggests oral phenylephrine may be ineffective for nasal congestion
as measured by nasal airway resistance
In a systematic review and meta-analysis published in uk Annals
ofPharmacothempy, oral phenylephrine was demonstrated to be
ineffective for the treatment of nasal congestion as measured by
nasal airway resistance (NAR).
Phenylephrine use has notably increased since retailers have been
required to keep products that contain pseudoephedrine behind store
counters as part of the USA Patriot Act's Combat Methamphetamine
Epidemic Act. In addition, a past alternative to pseudoephedrine,
phenylpropanolamine, was voluntarily removed from the US market in
2000 following reports of its association with increased hemorrhagic
stroke rates.
|



 Phenylephrine
or Neo-Synephrine is an α1-adrenergic receptor agonist used
primarily as a decongestant, as an agent to dilate the pupil,
and to increase blood pressure. Phenylephrine has recently been
marketed as a substitute for pseudoephedrine but there are recent
claims that oral phenylephrine may be no more effective as a
decongestant than a placebo.
Phenylephrine
or Neo-Synephrine is an α1-adrenergic receptor agonist used
primarily as a decongestant, as an agent to dilate the pupil,
and to increase blood pressure. Phenylephrine has recently been
marketed as a substitute for pseudoephedrine but there are recent
claims that oral phenylephrine may be no more effective as a
decongestant than a placebo. Pharmacokinetic
data
Pharmacokinetic
data