|
HOME >>
API >>
API List 3 >>
Theophylline

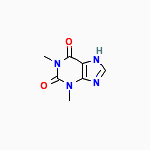
Cas No. [58-55-9]
Leading manufacturers of Theophylline and its
derivatives in India.
|
Name |
: |
Theophylline
 |
| CAS # |
: |
[58-55-9] |
| End Use |
: |
Bronchodilator |
IUPAC Name:
1,3-dimethyl-7H-purine-2,6-dione | CAS Registry Number:
58-55-9
Synonyms: theophylline, Elixophyllin, Theophyllin, Theolair,
Theocin, Nuelin, Synophylate, Bronkodyl, Aerolate, Theovent, Respbid,
Theobid, Uniphyl, 1,3-Dimethylxanthine, Pseudotheophylline,
aminophylline, Slo-phyllin, Elixophylline, Liquophylline,
Armophylline
| Molecular Formula: |
C7H8N4O2 |
Molecular Weight: |
180.164020 [g/mol] |
| H-Bond Donor: |
1 |
H-Bond Acceptor: |
4 |
A methyl xanthine derivative from tea with diuretic,
smooth muscle relaxant, bronchial dilation, cardiac and central
nervous system stimulant activities. Theophylline inhibits the
3',5'-CYCLIC NUCLEOTIDE PHOSPHODIESTERASE that degrades CYCLIC AMP
thus potentiates the actions of agents that act through ADENYLATE
CYCLASE and cyclic AM
![Cas No. [58-55-9]](api%20image/Theophylline/Theophylline56.gif)
DRUG CLASS AND MECHANISM:
Theophylline belongs to a class of medications called
bronchodilators, used in treating
asthma and other airway diseases.
Asthma is a breathing problem involving narrowing of the
airways. Airways are breathing passages that allow air to move in
and out of the lungs. Airways can be narrowed due to accumulation of
mucus, spasm of the muscles that surround these airways, or swelling
of the lining of the airways. Airway narrowing leads to symptoms of
shortness of breath, wheezing, cough and congestion. The narrowed
airways can open either spontaneously or from medications.
Medications that open airways are called bronchodilators.
Theophylline opens airways by relaxing the smooth muscles in the
walls of the airways. Theophylline can also be helpful in patients
with
emphysema and
chronic bronchitis when their symptoms are partially related to
reversible airway narrowing. Theophylline also strengthens right
heart function and diaphragm movement.
The main actions of theophylline
involve:
• relaxing bronchial
smooth muscle
• increasing heart
muscle contractility and efficiency: positive inotropic
• increasing heart
rate: positive chronotropic
• increasing blood
pressure
• increasing renal
blood flow
• some
anti-inflammatory effects
• central nervous
system stimulatory effect mainly on the medullary respiratory
center.
History
Theophylline was first extracted
from tea leaves and chemically identified around 1888 by the German
biologist Albrecht Kossel.Just seven years after its discovery, a
chemical synthesis starting with 1,3-dimethyluric acid was described
by Emil Fischer and Lorenz Ach. The Traube synthesis, an alternative
method to synthesize Theophylline has been introduced in 1900 by
another German scientist, Wilhelm Traube. Theophylline's first
clinical use came in 1902 as diuretic.It took additional 20 years
until its first description in asthma treatment.
Pharmacokinetics
Absorption
Bioavailability is 100%.
However, taking the drug late in the evening may slow the absorption
process, without affecting the bioavailability. Taking the drug
after a meal high in fat content will also slow down the absorption
process, without affecting the bioavailability.
Distribution
Theophylline is distributed in
the extracellular fluid, in the placenta, in the mother's milk and
in the central nervous system. The volume of distribution is 0.5
L/kg. The protein binding is 40%. The volume of distribution may
increase in neonates and those suffering from cirrhosis or
malnutrition, whereas the volume of distribution may decrease in
those suffering from obesity.
Metabolism
Theophylline is metabolized
extensively in the liver (up to 70%). It undergoes N-demethylation
via cytochrome P450 1A2. It is metabolized by parallel first order
and Michaelis-Menten pathways. Metabolism may become saturated
(non-linear), even within the therapeutic range. Small dose
increases may result in disproportionately large increases in serum
concentration. Methylation in Theophylline is also important in the
infant population. Smokers and people with hepatic (liver)
impairment metabolize it differently.
Elimination
Theophylline is excreted
unchanged in the urine (up to 10%). Clearance of the drug is
increased in these conditions: children 1 to 12, teenagers 12 to 16,
adult smokers, elderly smokers, cystic fibrosis, hyperthyroidism.
Clearance of the drug is decreased in these conditions: elderly,
acute congestive heart failure, cirrhosis, hypothyroidism and
febrile viral illness.
The elimination half-life
varies: 30 hours for premature neonates, 24 hours for neonates, 3.5
hours for children ages 1 to 9, 8 hours for adult non-smokers, 5
hours for adult smokers, 24 hours for those with hepatic impairment,
12 hours for those with congestive heart failure NYHA class I-II, 24
hours for those with congestive heart failure NYHA class III-IV, 12
hours for the elderly.
Indications
The main therapeutic uses of
theophylline are aimed at:
• chronic obstructive
pulmonary disease (COPD)
• asthma
• infant apnea
Uses Under Investigation
A clinical study reported in
2008 that theophylline was helpful in improving the sense of smell
in study subjects with anosmia.
Side-effects
The use of theophylline is
complicated by the fact that it interacts with various drugs,
chiefly cimetidine and phenytoin, and that it has a narrow
therapeutic index, so its use must be monitored to avoid toxicity.
It can also cause nausea, diarrhea, increase in heart rate,
arrhythmias, and CNS excitation (headaches, insomnia, irritability,
dizziness and lightheadedness). Its toxicity is increased by
erythromycin, cimetidine, and fluoroquinolones, such as "cipro"
(ciprofloxacin). It can reach toxic levels when taken with fatty
meals, an effect called dose dumping.
>>
New Product
Introduced :
Oseltamivir
Phosphate,
Phenyl Propanolamine,
Phenylephrine,
Etafedrine
|






![Cas No. [58-55-9]](api%20image/Theophylline/Theophylline56.gif)












