|
HOME >>
Chemicals
>>
Chemicals List 2 >> Para Anisyl Bromide
Para Anisyl Bromide
CAS Registry Number: 104-92-7
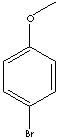
IUPAC Name: 1-bromo-4-methoxybenzene
Synonyms: p-Bromoanisole, Anisyl bromide, p-Bromanisole, Anisole, p-bromo-,
p-Anisyl bromide, 1-Bromo-4-methoxybenzene, 4-BROMOANISOLE, p-Methoxybromobenzene,
4-Methoxybromobenzene, p-Methoxyphenyl bromide, 4-Methoxyphenyl
bromide, Benzene, 1-bromo-4-methoxy-, 4-bromomethoxybenzene,
4-Methoxy-1-bromobenzene, p-Bromophenyl methyl ether,
B56501_ALDRICH, NSC 8042, EINECS 203-252-1, NSC8042, ZINC00404306
Molecular Formula: C7H7BrO
Molecular Weight: 187.033880
PRODUCT IDENTIFICATION
CAS NO 104-92-7
p-BROMOANISOLE
EINECS NO. 203-252-1
FORMULA BrC6H4OCH3
MOL WT. 187.04
H.S. CODEDERIVATION
TOXICITY
Oral, mouse LD50: 2200 mg/kg
SYNONYMS
4-Bromoanisole; 4-Bromo-1-methoxybenzene;
p-Anisyl bromide; 1-Bromo-4-methoxybenzene; p-Bromophenyl methyl
ether; p-Methoxybromobenzene;
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
PHYSICAL STATE
clear to slightly yellow liquid
MELTING POINT 9 - 11 C
BOILING POINT 223 C
SPECIFIC GRAVITY 1.494
SOLUBILITY IN WATER immiscible
pH
VAPOR DENSITY 4.28
AUTOIGNITION NFPA RARING
Health: 1; Flammability: 0; Reactivity: 0
REFRACTIVE INDEX 1.563
FLASH POINT 94 C
APPLICATIONS
Ether is any of a number of organic compounds characterized by an
oxygen atom joined with single bonds by two carbon atoms that are
part of hydrocarbon groups. The general formula is R-O-R', where R
and R' are alkyl or aromatic groups. Ethers are formed by the
condensation of two alcohols by heating with sulfuric acid; the
reaction is one of dehydration. Ethers can be prepared from alkyl
halide reacted with metallic alkoxide (called Williamson synthesis).
Ethers are similar to alcohols but are generally less dense, less
soluble in water, and have lower boiling points. They are relatively
unreactive, which makes them valuable solvents. But ethers will be
cleaved at high temperatures by concentrated hydrogen halides.
Ethers have relatively low boiling point compare to alkanes as they
don't form hydrogen bonds each other.
Ethers are more lipophilic than esters [R-C(=O)-O-R']or amides
[RCO-NH2]. Ethers are widely used as solvents for various organic
reactions because they are relatively the least reactive among
common organic compounds except alkanes and fluorocarbons. The
common reaction of ethers is cleavage of the C–O bond by strong
acids either in linear chain or cyclic structure. Ethers in which
oxygen is bonded to primary and secondary alkyl groups can form
peroxide compounds in the presence of gaseous oxygen due to two
unpaired electrons in oxygen. Ethers can act as Lewis bases in
chemical reactions. Commonly, ethers are named simply in listing the
alkyl groups in alphabetical order or alkane order such as ethyl
methyl ether or methyl ethyl ether, which is methoxyethane in IUPAC
nomenclature ( the formula of "alkoxyalkane" ).
When ether is a parts of complex molecule or aromatic derivatives,
it is described as an alkoxy substituent such as methoxybenzene (
trivial name is anisole). The methoxy prefix indicates the function
methyl group joined by single bonds to an oxygen atom, with the
general formula -O-CH3. Cyclic ethers have ring structure where the
oxygen has become part of the ring. The term of epoxide indicate
three membered cyclic ether (also called oxirane) in which an oxygen
atom is joined to each of two carbon atoms that are already bonded
to each other; four membered cyclic ether is called oxetane; five
membered cyclic ether, furan (or oxolane); six membered cyclic
ether, pyran (also called oxane) respectively. Their unhindered
oxygen atom carries two unshared pairs of electrons - a structure
which favors the formation of coordination complexes and the
solvation of cations. Cyclic ethers are used as important solvents,
as chemical intermediate and as monomer for ring-opening
polymerization. Crown Ether is a macrocyclic polyether whose
structure contains hydrogen, carbon and oxygen atoms. Each oxygen
atoms are confined between two carbon atoms and exhibits a
conformation with a hole (accordingly called "crown"). Anisole is
one of the simplest aromatic compound to which ether group is
linked. But it is different with aromatic compounds like furan where
the oxygen is a part of the ring.
Anisole, C6H5OCH3 (methyl phenyl ether), is a clear liquid that is
soluble in ether and alcohol; insoluble in water; boiling point 155
C. Anisole and its derivatives are used as solvents and in
perfumery. Anisole can be obtained from anise seed. Anisic acid, p-methoxybenzoic
acid, is a part of cresol class antiseptic compounds. It is also
used as an insect repellent and ovicide. Anisole, anisic acid, and
their derivatives are also widely used in chemical reaction as
intermediates to obtain target materials such as dyes,
pharmaceuticals, perfumes, photoinitiators and agrochemicals.
| |
|
Note /Government
Notification: These chemicals are designated as those that are
used in the manufacture of the controlled substances and are
important to the manufacture of the substances. For any (Control
Substance) products Import and Export *** subjected to your
country government laws /control substance ACT.
Information: The information on this web page is provided to
help you to work safely, but it is intended to be an overview of
hazards, not a replacement for a full Material Safety Data Sheet
(MSDS). MSDS forms can be downloaded from the web sites of many
chemical suppliers. ,also that the information on the PTCL
Safety web site, where this page was hosted, has been copied
onto many other sites, often without permission. If you have any
doubts about the veracity of the information that you are
viewing, or have any queries, please check the URL that your web
browser displays for this page. If the URL begins "www.tajapi.com/www/Denatonium
Benzoate.htm/" the page is maintained by the Safety Officer in
Physical Chemistry at Oxford University. If not, this page is a
copy made by some other person and we have no responsibility for
it.
The Controlled Substances Act (CSA) was enacted into law by the
Congress of the United States as Title II of the Comprehensive
Drug Abuse Prevention and Control Act of 1970.[1] The CSA is the
federal U.S. drug policy under which the manufacture,
importation, possession, use and distribution of certain
substances is regulated. The Act also served as the national
implementing legislation for the Single Convention on Narcotic
Drugs |
|
|
 |



