|
HOME >>
Chemicals
>>
Chemicals List 1 >> Phenylpropanolamine
Phenylpropanolamine
CAS number 14838-15-4
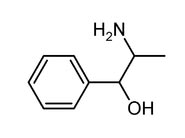
Identifiers
Systematic (IUPAC) name (1R,2S)-2-amino-1-phenyl-propan-1-ol
Identifiers
CAS number 14838-15-4
ATC code R01BA01
PubChem 26934
DrugBank APRD00457
ChemSpider 25082
Chemical data
Formula C9H13NO
Mol. mass 151.206 g/mol
Pharmacokinetic data
Half life 2.1 to 3.4 hours.
Therapeutic considerations
Routes Oral
Phenylpropanolamineis a drug ingredient of the phenethylamine family
used as a decongestantin prescription and nonprescription (over the
counter) cough and cold, and sinus remedies, and some combination
allergy medications. It is also present in an appetite suppressant.
Phenylpropanolamine (PPA) is used to treat nasal congestion
associated with the common cold, allergies, hay fever, or other
respiratory illnesses (e.g., rhinitis, sinusitis). It has also been
used as a nonprescription diet-aid for weight loss.
The Food and Drug Administration (FDA) is taking steps to remove
phenylpropanolamine (PPA) from all drug products and has requested
that all drug companies discontinue marketing products containing
PPA. In addition, FDA has issued a public health advisory concerning
phenylpropanolamine.
This drug is an ingredient that was used in many over-the-counter
(OTC) and prescription cough and cold medications as a decongestant
and in OTC weight loss products.
In response to the request made by FDA in November 2000, many
companies have voluntarily reformulated and are continuing to
reformulate their products to exclude PPA while FDA proceeds with
the regulatory process necessary to remove PPA from the market.
Use caution when driving, operating machinery, or performing other
hazardous activities. Phenylpropanolamine may cause dizziness or
drowsiness. If you experience dizziness or drowsiness, avoid these
activities. Never take this medication in larger doses or more often
than is recommended. Too much phenylpropanolamine could be very
harmful.
Before taking this medication, tell your doctor if you have
* high blood pressure;
* any type of heart disease, hardening of the arteries, or irregular
heartbeat;
* thyroid problems;
* diabetes;
* glaucoma or increased pressure in your eye;
* an enlarged prostate or difficulty urinating; or
* liver or kidney disease.
Use caution when driving, operating machinery, or performing other
hazardous activities. Phenylpropanolamine may cause dizziness or
drowsiness.
If you experience dizziness or drowsiness, avoid these activities.
Never take this medication in larger doses or more often than is
recommended. Too much phenylpropanolamine could be very harmful.
| |
|
Note /Government
Notification: These chemicals are designated as those that are
used in the manufacture of the controlled substances and are
important to the manufacture of the substances. For any (Control
Substance) products Import and Export *** subjected to your
country government laws /control substance ACT.
Information: The information on this web page is provided to
help you to work safely, but it is intended to be an overview of
hazards, not a replacement for a full Material Safety Data Sheet
(MSDS). MSDS forms can be downloaded from the web sites of many
chemical suppliers. ,also that the information on the PTCL
Safety web site, where this page was hosted, has been copied
onto many other sites, often without permission. If you have any
doubts about the veracity of the information that you are
viewing, or have any queries, please check the URL that your web
browser displays for this page. If the URL begins "www.tajapi.com/www/Denatonium
Benzoate.htm/" the page is maintained by the Safety Officer in
Physical Chemistry at Oxford University. If not, this page is a
copy made by some other person and we have no responsibility for
it.
The Controlled Substances Act (CSA) was enacted into law by the
Congress of the United States as Title II of the Comprehensive
Drug Abuse Prevention and Control Act of 1970.[1] The CSA is the
federal U.S. drug policy under which the manufacture,
importation, possession, use and distribution of certain
substances is regulated. The Act also served as the national
implementing legislation for the Single Convention on Narcotic
Drugs |
|
|
 |



