|
HOME >>
Chemicals
>>
Chemicals List 1 >> Safrole
Safrole
CAS number 94-59-7
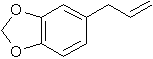
Safrole IUPAC name 5-(2-Propenyl)-1,3-benzodioxole
Other names Shikimol 5-Allylbenzo[d][1,3]dioxole
Properties
Molar mass 162.19 g/mol
Molecular Formula C10H10O2
Molecular Weight 162.19
Density 1.096 g/cm3
Melting point 11 °C
Boiling point 232-234 °C
Safrole is a colorless or slightly yellow oily liquid. It is
typically extracted from the root-bark or the fruit of sassafras
plants in the form of sassafras oil, or synthesized from other
related methylenedioxy compounds.
It is the principal component of brown camphor oil, and is found in
small amounts in a wide variety of plants, where it functions as a
natural pesticide.
Safrole is an ingredient used to manufacture Ecstasy, a psychoactive
drug that affects the brain's use of the naturally-produced chemical
serotonin, which regulates mood and aggression.The DEA issued a
public advisory in May 2003 warning chemical distributors about the
drug-making potential of safrole, which is a substance naturally
found in the oils from sassafras and camphor trees.
The Drug Enforcement Administration and state/local law enforcement
authorities have noted an alarming trend involving illicit MDMA
production. MDMA (Ecstasy) is a major drug problem.
All businesses engaged in the sale of safrole and essential oils
rich in safrole, such as, sassafras oil, "brown" camphor oil or
camphor oil 1.070, also referred to as Chinese sassafras oil, should
be aware of the use of these products by clandestine MDMA laboratory
operators.
Criminal elements routinely use these List I chemicals in the
illicit production of MDMA. DEA is aware that these criminals are
searching, often over the Internet, for sources of supply for these
List I chemicals.
This notice is being provided to:
Make you aware that safrole and essential oils rich in safrole, such
as sassafras oil, and brown camphor oil or camphor oil 1.070, are
List I chemicals;
Remind all distributors of these products that they must be
registered with DEA, obtain identifying information from their
customers, keep records, report suspicious orders to DEA, and "know
their customer," to avoid unwittingly becoming a supplier to a
clandestine MDMA laboratory;
Safrole was once widely used as a food additive in root beer,
sassafras tea, and other common goods. However, the FDA barred the
use of safrole after it was shown to be mildly carcinogenic.
Today, safrole is used for making soap and perfumes, aromatheraputic
agents, and the insecticide heliotropin (piperonal butoxide). More
recently, safrole has been used as the main precursor for the
clandestine manufacture of MDMA (Ecstasy) and MDEA (Eve). The
standard synthetic procedure for the production of MDMA from safrole
is via isomerization in the presence of a strong base to isosafrole.
This is then oxidised to 3,4-methylenedioxy phenyl-2-propanone.
Finally a reductive amination with either methylamine (to make MDMA)
or ethylamine (to make MDEA).
It is nearly impossible to obtain large quantities of safrole and/or
sassafras oil without arousing the suspicion of law enforcement, as
Safrole is currently a List I chemical.
Carcinogenicity
Safrole is shown to be a weak carcinogen in rats. It naturally
occurs in a variety of spices such as basil, cinnamon, nutmeg, and
pepper. In that role safrole is believed, although not proven, to
make a small but measurable contribution to the overall incidence of
human cancerToday, safrole is also banned for use in soap and
perfumes by the International Fragrance Association.
| |
|
Note /Government
Notification: These chemicals are designated as those that are
used in the manufacture of the controlled substances and are
important to the manufacture of the substances. For any (Control
Substance) products Import and Export *** subjected to your
country government laws /control substance ACT.
Information: The information on this web page is provided to
help you to work safely, but it is intended to be an overview of
hazards, not a replacement for a full Material Safety Data Sheet
(MSDS). MSDS forms can be downloaded from the web sites of many
chemical suppliers. ,also that the information on the PTCL
Safety web site, where this page was hosted, has been copied
onto many other sites, often without permission. If you have any
doubts about the veracity of the information that you are
viewing, or have any queries, please check the URL that your web
browser displays for this page. If the URL begins "www.tajapi.com/www/Denatonium
Benzoate.htm/" the page is maintained by the Safety Officer in
Physical Chemistry at Oxford University. If not, this page is a
copy made by some other person and we have no responsibility for
it.
The Controlled Substances Act (CSA) was enacted into law by the
Congress of the United States as Title II of the Comprehensive
Drug Abuse Prevention and Control Act of 1970.[1] The CSA is the
federal U.S. drug policy under which the manufacture,
importation, possession, use and distribution of certain
substances is regulated. The Act also served as the national
implementing legislation for the Single Convention on Narcotic
Drugs |
|
|
 |



